
Since OCH 3 is a more activating substituent than CH 3 (i.e. Here is a useful (but not comprehensive) ranking of activating / deactivating groups: 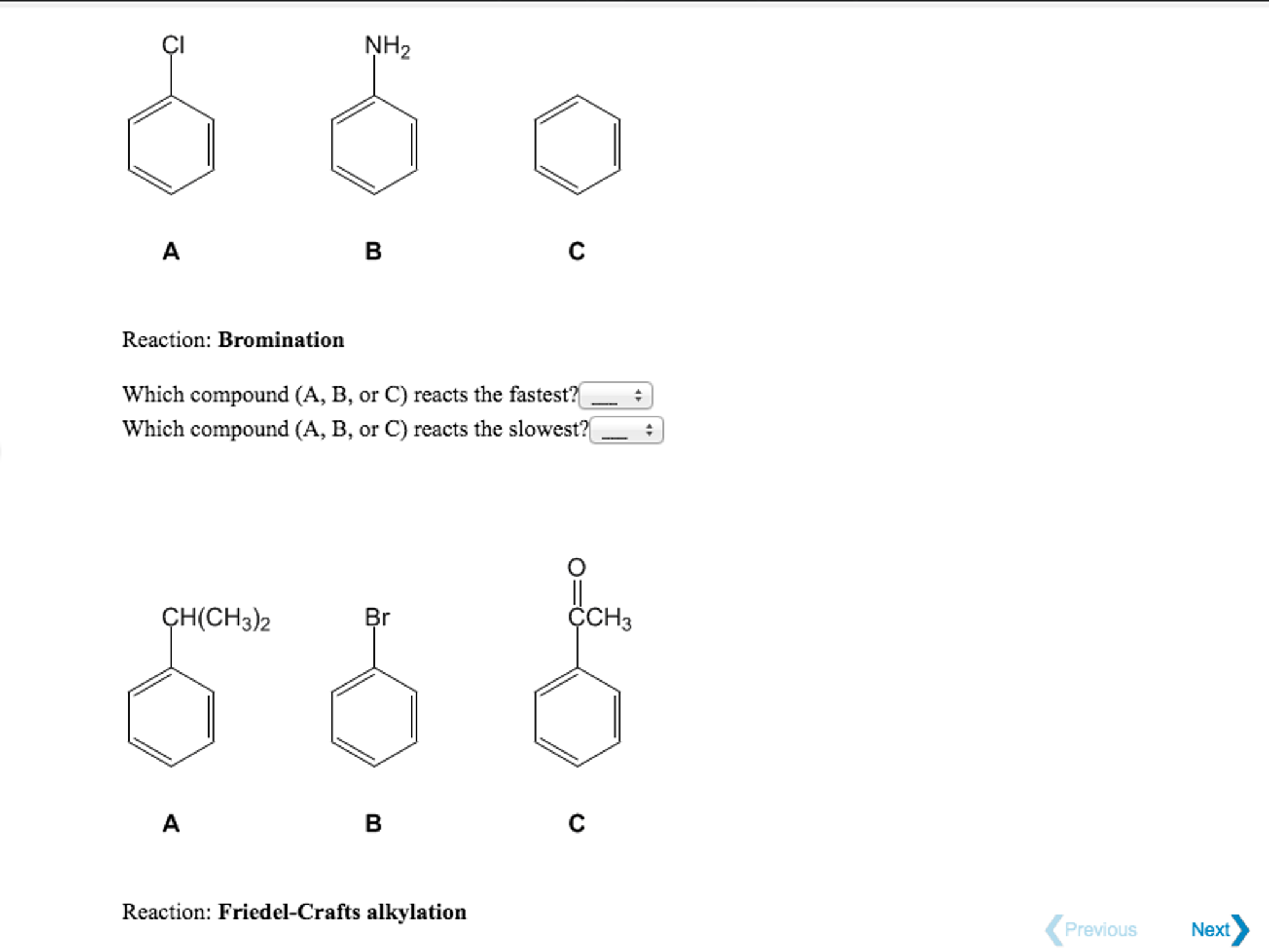
(that is, the more activating substituent “wins”) Rule #1: When two or more substituents are present on an aromatic ring, the directing group will be the most activating substituent. The tricky part is that they each direct to different carbons. Here there are two o-, p- directors: –OCH 3 and –CH 3. Let’s look at a slightly more ambiguous example: p-methylanisole. When Two Or More Substituents Are Present, The Directing Group W ill Be The Most A ctivating Substituent. This gives us the product 2-chloro-1- methoxy-4-nitrobenzene, which indeed is the major product.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
